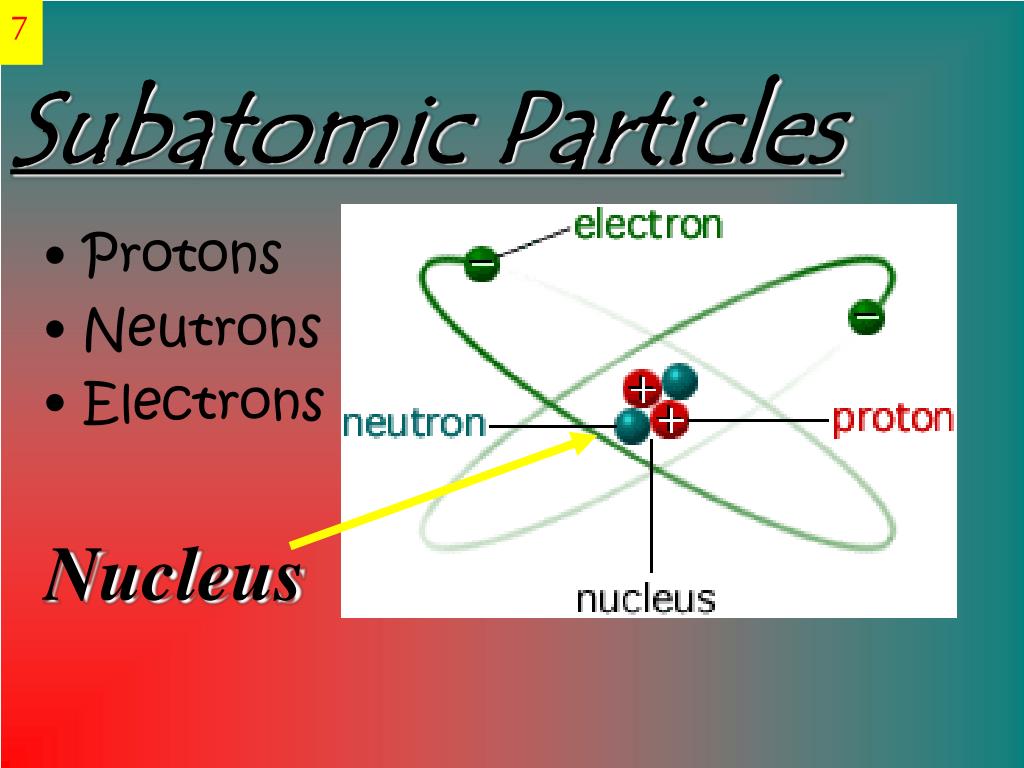
In a quantum system, it is given by the root mean square of the eigenvalue of the coordinate operator in its ground state. The size of a subatomic object must be carefully defined. And to be able to describe the valley of stability of nuclei as a function of the number of protons and neutrons. To know general facts about the spin of nuclei. The goals for you are to know how nuclear size is measured and what the results are. In this second video, we will summarize what is known about the size and the spin of nuclei. During this second module, we deal with nuclear physics and its applications. And the last module introduces you to two mysterious components of the Universe, namely Dark Matter and Dark Energy. Module 7 deals with our ways to search for new phenomena. The modules 4 to 6 go into more depth about matter and forces as described by the standard model of particle physics. Following the first one which introduces our subject, the modules 2 (nuclear physics)Īnd 3 (accelerators and detectors) are rather self contained and can be studied separately. The course is structured in eight modules. What can one learn from particle physics concerning astrophysics and the Universe as a whole? How does one search for new phenomena beyond the known ones? What is the mass of objects at the subatomic level and how does the Higgs boson intervene? How do weak interactions work and why are they so special? How do strong interactions work and why are they difficult to understand? How do electromagnetic interactions work and how can one use them? What does one learn from particle reactions at high energies and particle decays? How does one accelerate and detect particles and measure their properties? What are the properties of atomic nuclei and how can one use them? What are the concepts of particle physics and how are they implemented? More specifically, the following questions are addressed: The nucleus shows a positive charge due to the net positive charge of protons in it.This course introduces you to subatomic physics, i.e.The nucleus of an atom is filled with tightly packed protons and neutrons. 
The mass of an atom depends on the mass of its nucleus.The central area or middle portion of an atom is called the nucleus of an atom.The scientist James Chadwick discovered the neutrons.The mass of neutrons is quite more than the mass of protons.The neutrons have zero charges on them.Neutrons are present inside the nucleus of an atom with protons.The subatomic particle of atoms containing no charge or neutral is called neutrons.The scientist Goldstein discovered the protons.. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
